|
The sodium absorption spectrum consists of two dark lines corresponding to 589 nm and 589.6 nm against a bright background. 
The dark line corresponds to the absorption of characteristic radiation by an atom of the element. If the beam emerging from flame is analyzed in the spectrometer, an absorption spectrum consisting of dark lines against a bright background is observed. Sodium will absorb the wavelength radiation of 589 nm and 589.6 nm. Allan Walsh in 1955.Įxample: When a beam of radiation is passed through a flame containing the atoms of an element in the ground state, the atom will absorb their characteristic wavelength for e.g. This elegant method was discovered by Sir. Hence AAS is an invaluable tool for quantitative and qualitative analysis of a large number of elements. The difference in intensity of incident radiation and emitted radiation gives the intensity of absorption which in turn gives the number of atoms in the sample at the ground state and hence forms the basis of quantitative analysis. LARK® 1.This absorption of characteristic radiation forms the basis of qualitative analysis of elements under study since every element absorbs its own characteristic radiation. More than 52+ Types of Lamps Ready stock. LARK® HCL can be used on Atomic Absorption Spectrometers manufactured by: Reliable quality so your expectations will always be met.Wide selection so LARK® can be your one source for all HCL’s.Enhanced sensitivity to produce great results even with difficult samples.Low operating current to extend the lifetime of lamps even further.  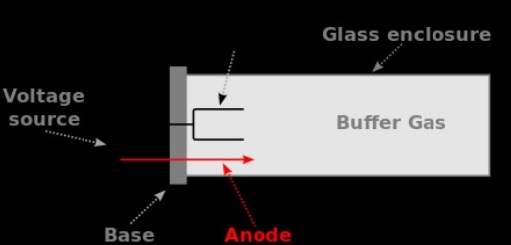
High-intensity light output resulting in low signal-to-noise ratio and excellent performance.Long life for economical, operator convenience.LARK®’s technical ability to produce hollow cathode lamps that meet the demanding specifications of instrument manufacturers is only surpassed by the company’s customer service and on-time delivery responsiveness. LARK® Hollow Cathode Lamps are manufactured in different sizes and with argon and/or neon gas fills to assure optimum results on a wide variety of instruments. Lamps for use with Smith-Hieftje background correction can also be offered. The range includes standard lamps and data-coded versions for Perkin Elmer and Thermo Fisher atomic absorption spectrometers. LARK® Hollow Cathode Lamps are available both for OEMs and as a replacement lamp by discerning users the world over. LARK® offers the widest selection of single- and multi element coded / non-coded in low and high-current, 37 mm and 50 mm lamps in the industry. They consist of a cathode made from the element of interest, an anode and an inert filler gas contained in a glass envelope. LARK® Hollow Cathode Lamps (HCL) are discharge lamps designed for use in Atomic Absorption (AA) instruments. The information from the built-in electronic lamp timer can be accessed easily from the AA Synergistic software of the Pinnacle Atomic Absorption Spectrometers. The larger the lamp, the greater the inert gas volume - and the longer the lamp lifetime. 
To prolong the life of a LARK® Hollow Cathode Lamp (HCL), we produce lamps with larger internal volume so that a greater supply of fill gas at optimum pressure is available. Every genuine LARK® lamp is designed for use with and tested on Perkin Elmer spectrometers to assure compatibility and the highest performance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
